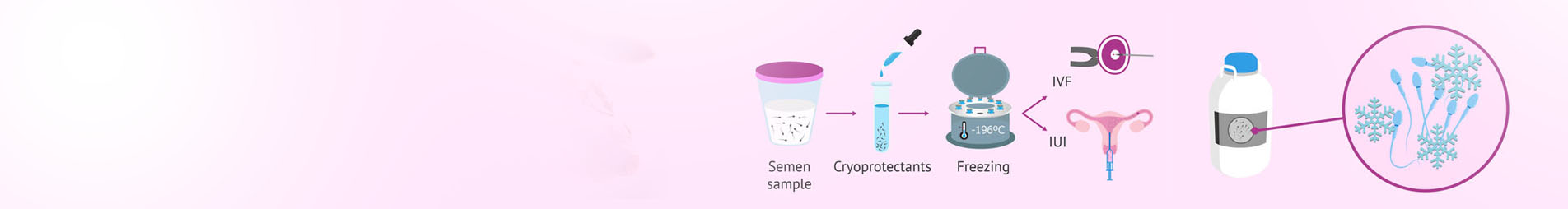
Cryopreservation or cryoconservation is a process where cells, whole tissues, or any other substances susceptible to damage caused by chemical reactivity or time are preserved by cooling to sub-zero temperatures. In embryology lab there are two techniques that used for freezing of sperm , egg, embryos and ovarian tissues . Slow Freezing and Vitrification